|
(Recall that the number of valence electrons is indicated by the position of the element in the periodic table. 
Add together the valence electrons from each atom. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Determine the total number of valence electrons in the molecule or ion. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. Neutral sodium atom on left has 11 protons and 11 electrons. 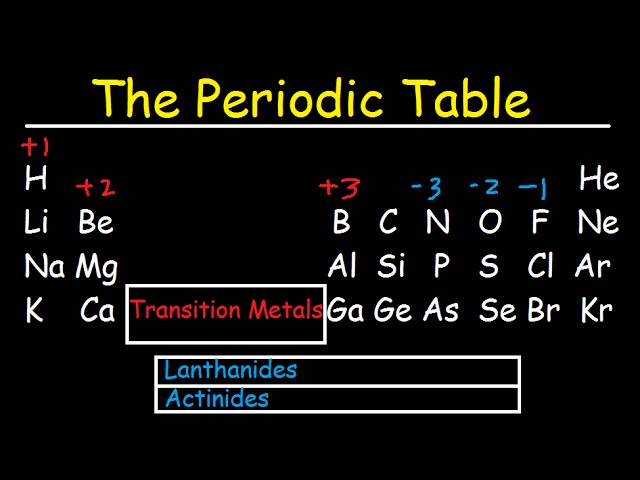
On the right, the sodium ion only has 10 electrons and a 1+ charge. On the left, a sodium atom has 11 electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). 1 is a graphical depiction of this process. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. We can see this general size trend in the following periodic table. 
Electronegativity decreases as we move down a column in the periodic table. Each element square contains all 118 of elements with the element number, symbol, name, atomic mass, and most common oxidation number. The two elements following argon in the periodic table are potassium, with a single 4s electron, and calcium, with two 4s electrons. Moving down a column in the periodic table, valence electrons are held less tightly because they get further from the nucleus. Reviewing the way of finding the charge on an atom may help, too. There are total 18 vertical columns on periodic table. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
